Zooming in on legumes offers hope for nitrogen pollution crisis
Angus Rae likes nothing more than peering through the confocal microscope at the ANU Research School of Biology. “I love that microscope,” he says. “It’s amazing, the enjoyment I get from it.”
Three years into his PhD, and Angus spends so much time looking inside plant cells that these “beautiful” images have even appeared in his dreams.
And while Angus focuses intently on a captivating microscopic world, he is actually working towards a solution for an environmental catastrophe occurring on a global scale.
“Nitrogen pollution is a real problem that doesn’t get enough attention in the news,” Angus says. Run-off from nitrogen fertilisers has contributed to more than 400 dead zones in the world’s oceans, and over half of the 120 million tonnes of synthetic nitrogen used globally on crops every year ends up washing into rivers.
And the problem isn’t limited to run-off, Angus says.
“In terms of climate change, you hear a lot about methane from cows, but a really big contributor to nitrous oxide emissions—which exacerbate climate change—is artificial nitrogen fertilisers. Also, we have to burn natural gases to produce these fertilisers, so that contributes to climate change too.
“But crops need nitrogen to grow, so we need a better source of nitrogen, and legumes may hold the key.”
This is where Angus comes in. And his microscope. Oh, and legumes, too.
“Beans, peas, and other plants in the legume family form a symbiosis—a mutually beneficial relationship—with special bacteria that lives inside their roots, and converts gaseous nitrogen into a usable form,” Angus explains.
“If we can understand how this symbiosis has evolved in legumes, and how it works, we should be able to make it happen in other plants and reduce our dependency on damaging fertilisers.”
It’s a bio-engineering solution to a human-made problem.
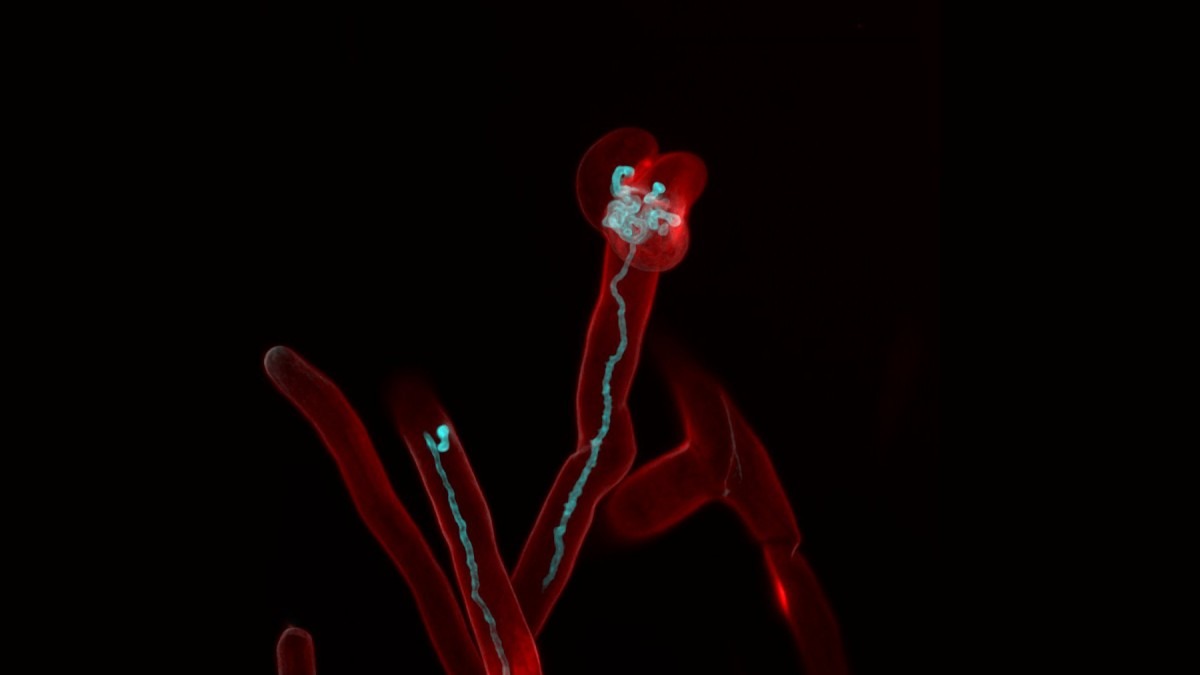
As part of his research, Angus has developed methods in advanced microscopy to try to work out exactly what is happening in legume roots, and how the bacteria “infect them, in a good way”: “We know that the plant essentially swallows the bacteria, by creating intestine-like tubes that pull them into the root.”
These twisted, tubes—“weird structures” called infection threads—are what keeps Angus glued to the microscope. He has already learnt how many there are per root, and per cell; how long they typically are; and where in cells they usually originate. But there’s more to know.
“The next step is to zoom in even further and work out the molecular signalling pathways that control the growth of infection threads,” he says, “so that we can potentially learn how to make them grow in other plants.”
In the lab, Angus doesn’t often stop and think about the future application of his research. That’s a long-term goal, he says, and he has short-term work to finish.
“Hopefully my research will make a contribution, and we already have some really cool results,” he says. “But I don’t actively think about that while I’m looking down a microscope.”
Instead, he says, he finds himself thinking more about the “complexity and beauty of plant design”.
“We tend to think of plants as fairly simple things compared to animals. But when you look at the biology of plants, it's amazing the way that they run.
“If you look at symbiosis, specifically, it’s just incredible that two such different organisms can work together in harmony, to benefit each other in this special way.”
Work in harmony with our internationally-recognised plant scientists as part of your degree at the ANU Research School of Biology.